1.Introduction to PVA
Polyvinyl alcohol (PVA) is an organic compound that appears as a white, flaky, flocculent, or powdery solid, odorless. It is soluble in water (above 95℃), slightly soluble in dimethyl sulfoxide, and insoluble in gasoline, kerosene, vegetable oil, benzene, toluene, dichloroethane, carbon tetrachloride, acetone, ethyl acetate, methanol, ethylene glycol, etc.
Polyvinyl alcohol is an important chemical raw material used in the manufacture of polyvinyl acetal, gasoline-resistant pipes and vinylon synthetic fibers, fabric treatment agents, emulsifiers, paper coatings, adhesives, and glues.
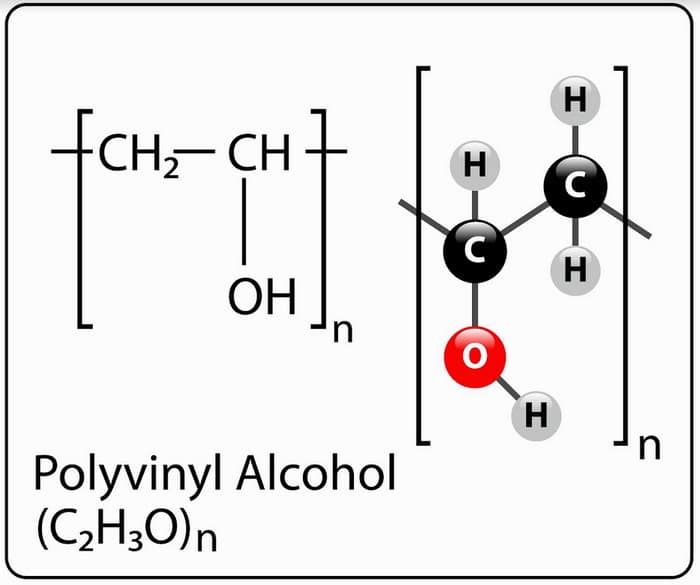
Chemical Formula of PVA
2.Properties of PVA
Physicochemical Properties of PVA
Polyvinyl alcohol is generally white or slightly yellow, appearing as a flocculent, granular, or powdery solid. It is non-toxic and odorless, with properties between plastics and rubber.
PVA solution turns deep blue upon contact with iodine solution; this color change disappears upon heating and reappears upon cooling. Due to the presence of numerous side hydroxyl groups on its molecular chain, it exhibits excellent water solubility, as well as good film-forming properties, adhesion, and emulsifying properties, along with excellent resistance to oils and solvents.
Its physical parameters are as follows:
Relative density (25℃/4℃): 1.27~1.31 (solid), 1.02 (10% solution)
Melting point: 230℃
Glass transition temperature: 75–85℃
When heated in air above 100℃, it gradually discolors and becomes brittle;
At 160–170℃, it undergoes dehydration and etherification, losing its solubility;
It begins to decompose at 200℃;
Above 250℃, it transforms into a polymer containing conjugated double bonds.
Other properties:
Refractive index: 1.49–1.52
Thermal conductivity: 0.2 W/(m·K)
Specific heat capacity: 1–5 J/(kg·K)
Resistivity: (3.1–3.8) × 10⁷ Ω·cm
Properties of PVA aqueous solutions
When the concentration of PVA aqueous solution is 1%–5%, its viscosity does not decrease significantly after prolonged standing or heating at room temperature, indicating no depolymerization.
As the solution concentration increases, the viscosity increases, and a supramolecular structure forms; gelation may occur after prolonged standing, but it can be restored to a homogeneous solution upon heating.
Viscosity Changes in PVA Aqueous Solutions
The viscosity of PVA aqueous solutions is affected by the type, concentration, and temperature:
PVA-1799: Has many hydroxyl groups, easily forms hydrogen bonds between molecules, readily cross-links, and its viscosity increases over time.
PVA-1788: Viscosity change is not significant, showing a basically linear relationship.
Stability of PVA Aqueous Solutions
PVA-1799 aqueous solutions increase in viscosity after standing, while PVA-1788 is relatively stable.
The opposite is true when boric acid or borax is added: As the degree of alcoholysis decreases, the required amount decreases, and stability decreases.
Medical Applications of PVA
PVA hydrogels possess the following characteristics:
Water content close to that of human tissue
Moderate elastic modulus
Low coefficient of friction
High mechanical strength
Porous structure
Good biocompatibility
Therefore, they have wide applications in the biomedical field, including:
Artificial cartilage
Artificial cornea
Artificial vitreous body
In ophthalmology:
Can be used to manufacture soft contact lenses
In pharmaceuticals:
Can be used to prepare sustained-release capsules
Furthermore, ionized polyvinyl alcohol sponges (i-PVA sponges) formed by introducing sodium polyacrylate side chains possess:
Good biocompatibility
Stable chemical properties
Excellent liquid absorption and hemostatic ability
Can be used for:
Microsurgery
Ophthalmic surgery
Cataract intraocular lens replacement surgery
Website: www.china-pva.net
Whatsapp: 0086-15380400285
E-mail: sales2@china-pva.net